Kerr Research Group
Russell Kerr

Russell G. Kerr
Department of Chemistry, and Department of Biomedical Sciences,
Atlantic Veterinary College, University of Prince Edward Island
Mailing address:
Russell Kerr
University of Prince Edward Island
Duffy Research Centre
550 University Avenue
Charlottetown PE C1A 4P3
Canada
Contacts:
Email: rkerr@upei.ca
Phone: (902) 566-0565
Office: Duffy Research Centre, Room 528
|
Biographical Information
B.Sc. University of Calgary 1982
Ph.D. University of Calgary 1987
Postdoctoral Fellow Stanford Univeristy (with C. Djerassi) 1987 - 1991
Academic Appointments:
1991-1996 Assistant Professor, Department of Chemistry, Florida Atlantic University.
1996-1998 Associate Professor, Department of Chemistry, Florida Atlantic University.
1998-2006 Professor, Department of Chemistry & Biochemistry, Florida Atlantic University.
1999-2006 Professor of Biomedical Sciences, Florida Atlantic University.
2000-2006 Director of Graduate Studies, Department of Chemistry & Biochemistry, Florida Atlantic University.
2003-2006 Director, Center of Excellence in Biomedical and Marine Biotechnology, Florida Atlantic University
2006- Professor & Canada Research Chair, Dept. of Chemistry, UPEI
2006- Professor & Canada Research Chair, Department of Biomedical Sciences, Atlantic Veterinary College, UPEI
Selected Awards:
2010 Lévesque Chair in Nutrisciences and Health
2006 Canada Research Chair (Tier 1) in Marine Natural Products
2004 Researcher of the Year, Florida Atlantic University
2003 Admitted to “Who's Who in Sciences Higher Education (WWSHE)”
2000 President’s Research Development Award (FAU)
2000 Elected as “Young Observer” for the U.S. National Committee for the International Union of Pure and Applied Chemistry (IUPAC)
1999 Cancer Drug Discovery study section, American Cancer Society (1999-2003)
1998 Charles Schmidt College of Science Distinguished Teacher of the Year
1997 Elected to Sigma Xi
1997 Phi Kappa Phi faculty award (FAU)
1997 Excellence in Undergraduate Teaching Award, Florida Atlantic University
1996 Henry Dreyfus Teacher-Scholar Award
1996 Researcher of the Year Award (Assistant Prof. level), FAU
1995 Teaching Incentive Program Award, FAU
1992 American Society of Pharmacognosy Research Initiation Award
Scientific Affiliations:
Chemical Institute of Canada
American Chemical Society
American Association for the Advancement of Science
American Society of Pharmacognosy
American Association for Cancer Research
Editorial Boards:
Letters in Organic Chemistry
Marine Drugs
The Open Organic Chemistry Journal
Research Letters in Organic Chemistry
|
Current Projects
 |
The impact of natural products on modern society is indisputable given their importance to medicine. As one example, 42% of anticancer and 47% of anti-infective agents introduced over the past 25 years are natural products. While remarkable discoveries have been made, the rate of natural product discovery has declined in recent years, profoundly affecting the development of new therapeutics and the related pharmaceutical and biotechnology industries. The general aim of many projects in our lab is to conduct research in natural products chemistry and associated microbiology and genetics to develop innovative methods to improve natural product discovery from microbes of marine origin. |
Pseudopterosins

Microbial Diversity
 |
Microbes (bacteria and fungi) are the source of all our natural products investigations and are obtained from a wide diversity of marine habitats. The marine environment represents an underexplored resource for microbial natural product discovery thus improving the likelihood of isolating new compounds. We are interested in projects directed at assessing microbial diversity using both culture independent and culture dependent methods. As is evident from the Field Work tab on this web page, we have on-going collection programs in Canada’s Arctic, Colombia, the Mediterranean, the Black Sea, The Bahamas as well as Atlantic Canada. |
Accessing Cryptic Natural Product Biosynthetic Pathways
 |
Through funding from NSERC, we have a number of student and postdoc projects that seek to develop new methods for the isolation of marine-derived microbes thus allowing access to new sources of natural products. Further, this research program aims to develop new methods to induce natural product production in microbial fermentation and seeks to develop a new chemical tool to efficiently identify new natural products from large sets of complex mixtures. Lastly, antimicrobial assays of relevance to veterinary medicine will be used to guide the isolation and characterization of new natural products. Overall, this research program will develop research methods that will enhance the rate of discovery of new bioactive natural products. |
 |
Marine Microbes as a New Source of Ingredients for the Personal Care Industry

Identification of novel enzymes involved in the breakdown of plant fibre to be used in the development of a ruminant feed additive
In collaboration with Nautilus and AB Vista, we are screening our marine microbial library for enzymes with applications as a feed additive for the ruminant feed industry which can increase fibre digestion. The overall goal of this project is to identify new novel xylanase, cellulase and endoglucanase activities and accessory enzymes (arabinofuranosidase, xylosidase and glucuronidases) which can be used to develop a new ruminant feed additive which will increase feed efficiency and productivity.
Research Environment
|
Kerr Lab Research Environment |
|
|
LOCATION:
|
|
|
The Kerr Lab is ideally located on the University of Prince Edward Island campus in Charlottetown, Prince Edward Island. The bacterial and chemical divisions of the lab are centred in the Regis and Joan Duffy Research Centre. The fungal division of the lab as well as our antibiosis/cell screening division are located in the North Annex of the Atlantic Veterinary College. Click here to access a full map of the UPEI campus in a new window. |
|
|
Regis and Joan Duffy Research Centre
The Regis and Joan Duffy Resaerch Centre, is home to researchers from UPEI, the National Research Council Institute for Nutrisciences and Health (NRC-INH), and Agri-Food Canada (AAFC). Here they share knowledge, lab space, and specialized equipment and tools as they explore how natural compounds can be used to prevent and treat disease in animals and humans, contributing to the island's growing bioscience cluster.
The Regis and Joan Duffy Research Centre boasts an open concept lab environment, which facilitates both the sharing of equipment and knowledge among the researchers. Both the Chemical and the Bacterial divisions of the Kerr Research lab are located on the fifth floor.
|
|
|
|
Atlantic Veterinary College, North Annex The North Annex expansion of the Atlantic Veterinary College (AVC) at the University of Prince Edward Island (UPEI) includes both office and laboratory space. Both the Fungal and the Bioassay (antimicrobial and cell based assay) divisions of the Kerr Research lab are located in our Biosafety Level 2 laboratory at 3rd flour in the North Annex of the AVC. Additional to our laboratory space, our group also has recourse to Multi User space and equipment that are made available from AVC.
|
KerrLab HRMS facility
|
|
|
|
The Kerr Marine Natural Products Lab has state-of-the-art equipment to conduct natural-product drug-discovery research. Two complimentary LC-HRMS instruments provides our lab with strong capabilities for the analysis of natural product extracts, the quantification of targeted metabolites in complex samples and the study of microbial secondary metabolomes using LC-HRMS assisted with statistical analyses. The Kerr Lab HRMS facility has recently developed an innovative methodology to dereplicate microbial strains by metabolomics approaches while providing the abilityto identify putative novel chemical entities as natural product discovery leads. http://www.sciencedirect.com/science/article/pii/S0003267013013378 |
|
|
The HRMS facility is available for consultation with all researchers on the UPEI campus as well as other universities to develop collaborative projects and assist researchers with mass spectrometry expertise. Mass spectrometry service is also available for academic institution and private companies on a fee-for-service basis (service rates). For more information please contact us, we would be pleased to help you with your sample analysis. |
|
|
|
System 1: LC-HRMS-PDA with an Orbitrap LTQ-Velos (Thermo Scientific) using either ESI or APCI ionization source is an ideal system for the quantification and the identification of natural products from pure samples or mixtures. This instrument can also perform tandem mass spectrometry MS(n) experiments and assist researchers with the structure elucidation of unknown molecules.
|
 |
System 2: LC-HRMS-ELSD with an Exactive mass spectrometer (Thermo Scientific) coupled to a universal evaporative light scattering detector (ELSD) is a perfect combination for the rapid screening of natural product extracts.
|
|
|
|
Contact us
Contact information |
||
|
Research Manager:
|
Doug Marchbank |
|
|
Mass Spec:
|
Hebelin Correa |
|
|
Location: |
Duffy Research Center, room 513 |
|
|
Mailing Address: Duffy Research Centre, rm 513 |
Telephone: 902-566-8051 |
|
Main Lab & Equipment
|
Lab Space and Equipment
|
|
|
The lab located on the fifth floor of the Duffy Research Centre forms the base of operations for bacteriology and chemistry oriented research in the Kerr Lab. The lab houses a wide selection of equipment need for microbiology, molecular biology and chemisty research. To support our natural products chemistry research the lab is equiped with eight fume hoods, four rotary evaporators, two vacuum evaporators (GeneVac), an automated flash chromatography system (Teledyne Combiflash Rf), a Thermo Scientific GCMS and four HPLC systems, one of which is connected to a mass spectrometer to enable mass-guided fractionation. The crown jewels of the lab are two Thermo Scientific Orbitrap high-resolution mass spectrometers (Exactive and Velos models), which are both connected to ultra-high performance liquid chromatography (UPLC) systems. For natural product structure elucidation a 600 MHz Bruker NMR (housed in the Duffy Research Centre) is made availabe via an agreement with Agriculture and Agri-food Canada. To support molecular biology research, the lab is equiped with two gradient-capable PCR thermocyclers, a UVP molecular imager, a Bio-Rad Duo-Flo FPLC system and a variety of electrophoresis supplies. Microbiology-related equipment includes three New Brunswick Innova 44 shaking incubators, a large ambient temperature platform shaker, two laminar flow hoods, a type II biosafety cabinent, a variety of microscopes and a Steris autoclave. The lab is also equiped with common general use equipment including multiple balances, centrifuges and stationary incubators, two ultralow temperature freezers, two dishwashers and a Millipore MilliQ deionized water system. A graphical represenation of the lab layout and pictures of various spaces and equipment are shown below.
 |
|
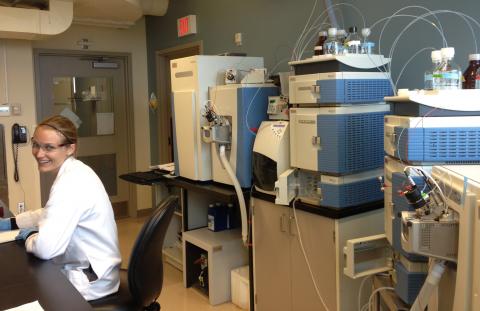
 Our HRMS instruments are the crown jewel of the lab! The instrument on the left is the Thermo Scientific Exactive HRMS. This instrument is coupeld to a UPLC chromatography system as well as a PDA detector and a universal evaporative light scattering detector (ELSD). This instrument provides an ideal platform for the rapid screening of natural product extracts. The instrument on the right is the Thermo Scientific Orbitrap LTQ-Velos. This instrument is also coupled with a UPLC and PDA detector. Using either ESI or APCI ionization sources, this instrument is ideal for the quantification and identification of natural products from pure samples or mixtures. This instrument can also perform tandem mass spectrometry MS(n) experiments to assist researchers with the structure elucidation of unknown molecules.
|
.jpg) Our GCMS instrument is a Thermo Scientific Focus GC, which is coupled with a Polaris Q quadrupole ion trap mass analyzer equiped with electron ionization source. This system is ideal for characterizing non-polar metabolites not compatable with reversed-phase LC.
|
 A recent addition to the lab is a Waters HPLC system coupled to a mass-guided fraction collector. The system consistes of two Waters HPLC pumps, a Waters 3100 single quadrupole mass spectrometer and a Waters 2767 fraction collector. This system is designed for both analytical and semi-preparative separations.
|
 A second Waters HPLC system is also available for analytical and semi-preparative separations. This system features a manual injector, a Waters 1525 binary HPLC pump, a Waters 2489 dual channel UV/Visible detector, a Waters 2424 ELSD and a Waters 2767 fraction collector.
|
 Our Thermo Scientific HPLC system features a Finnigan Surveyor Autosampler Plus, LC Pump Plus, and PDA Plus detector. It is also conntected to a SEDEX 60 LT ELSD. The system is designed for both analytical and semi-preparative separations.
|
 The latest model in medium-pressure chromatography from Teledyne Isco, the CombiFlash RF, is compact and user-friendly. The system is equipped with radio frequency identification (RFID) technology to allow for automation of parameters for purification and fraction collection.
|
 Evaporation is a common occurence in the purification of any natural product. To facilitate rapid evaporation of samples, the lab is equiped with four rotary evaporators (top photo) and two GenVac vacuum evaporators. The GeneVacs are ideal for drug discovery applications as the GeneVac EZ-2 Plus is capable of evaporating all commonly used solvents and most acids quickly and effectively.
|
 To facilitate bacterial fermentation, the lab is equipped with four shakers. One is a large ambient temperature platform shaker (bottom photo). The other three (top photo) are temperature controlled Innova 44 shaker incubators. One of these is a refridgerated unit, which allows us to explore the effect of temperature on natural product production.
|
 The molecular biology room (Rm 517) contains two gradient-capable thermal cyclers (Eppendorf and Bio-Rad), electrophoresis equipment, an incubator and a laminar flow hood.
|
 To support molecular biology activities the lab is equiped with a Bio-Rad Duo-Flo FPLC (top photo) system, which is used to purify proteins and other biomoleculres. Molecular imaging needs are met by the UVP BioSpectrum imaging system (bottom photo).
|
 A multitude of equipment is avialable for general use in the lab including (clockwise from top right) a compound microscope, a -80oC freezer, a Beckman Coulter Allegra 25R centrifuge, a freeze dryer and type II biosafety cabinet.
|
 Room 509 is a common area that houses the autoclave, as well as the dish washers and the Millipore MilliQ deionized water system.
|
Fungal and Bioassay Divisions at the AVC
|
Lab Space and Equipment
|
|
|
This laboratory space is focused upon fungal isolation, fermentation and extraction in addition to the preparation and evaluation of antimicrobial assays. Several mulit-user locations are also available on the 3rd floor of the North Annex, where we carry out media preparation, PCR amplification, and cell culture work (in a dedicated cell culture lab).
|
|
 Various incubators allow for culturing pathogens for antimicrobial assays at various temperatures.
|
 Biological Safety Cabinet used for working in a sterile environment
|
 Temperature controlled shaking incubator where we can carry out shaken fermentations of various volumes ranging from 20 mL - 2 L.
|
 Temeperature controlled standing incubator where we culture our fungi and carry out static fermentations.
|
 Chemistry work station: fumehood and lab bench.
|
 Various tools are available for the extraction and processing of fungal fermentations.
|
 Microscopy work station: Disecting scope and compound microscope used for morphological identification.
|
 Shaking Incubator used for culturing seed inoculum for fermenations and assays.
|
|
Vacuum concentrator (SpeedVac) used for extract evaporation when generating mother/daughter plates for antimicrobial assays. |
Spectrophotometer/Fluorometer and stacking robot used for evaluating results from 96 well assay plates. |
Various bench spaces are available to carry out routine lab activities such as DNA extraction and assay plate preparation. |
 |
Lab Members
 |
We try to get together as a group at least once a month to enjoy activities outside the lab. The Island is perfect for a variety of seasonal extracurricular events, such as skating, skiing, swimming, scuba diving, and of course, the occasional barbeque. Please click here or on the picture for some descriptions and pictures of our most recent group outings! | |||
|
Research and Office Staff |
||||
 Russ Kerr
Professor
Phone: 902-566-0565
|
Brad Haltli
Research Manager
Phone: 902-418-5125
|
|
||
| Postdoctoral Researchers | ||||
|
|
|
|||
| Graduate Students | ||||

Logan MacIntyre
PhD Student
|
 Anna Kuznetsova PhD Student
|
 Vernon Ptycia-Lamky MSc Student
|
Hope Igboeli
PhD Student
|
|
 Zach Maw
PhD Student
|
|
|
||
|
|
|
|||
Member information
Learn more about members of the Marine Natural Product Laboratory
Brad Haltli
1. Tangerina M.P., Correa H., Haltli B., Vilegas W., Kerr R.G. Bioprospecting from cultivable bacterial communities of marine sediment and invertebrates from the underexplored Ubatuba region of Brazil. Arch Microbiol. 2017 199: 155-169.
2. McCauley E.P., Haltli B., Correa H., Kerr R.G. Spatial and temporal investigation of the microbiome of the Caribbean octocoral Erythropodium caribaeorum. FEMS Micro Ecol. 2016 92: pii: fiw147. doi: 10.1093/femsec/fiw147.
3. Robertson V.*, Haltli B.*, Overy D., Kerr R.G. Highly variable bacterial communities associated with the octocoral Antillogorgia elisabethae. Microorganisms 2016 4: pii:E23. doi: 10.3390/microorganisms 4030023.
4. Pastrana-Camacho N., Suárez Z., Acosta-González A., Arango C., Haltli B., Correa H., Kerr R.G., Duque C., Diaz L.E. Bioprospecting for culturable actinobacteria with antimicrobial properties isolated from rivers in Colombian Orinoquia. Trop J Pharm Res. 2016 15: 1259-1265.
5. Sommer B., Overy D.P., Haltli B., Kerr R.G. Secreted lipases from Malassezia globosa: recombinant expression and determination of their substrate specificities. Microbiology 2016 162: 1069-79.
6. Herzog B., Overy D.P., Haltli B., Kerr R.G. Discovery of keratinases using bacteria isolated from marine environments. Syst Appl Microbiol. 2016 39: 49-57.
7. Arens J.C., Haltli B., Kerr R.G. Draft Genome Sequence of Kitasatospora griseola Strain MF730-N6, a Bafilomycin, Terpentecin, and Satosporin Producer. Genome Announc. 2015 3: e00208-15.
8. McCauley E.P., Haltli B., Kerr R.G. Description of Pseudobacteriovorax antillogorgiicola gen. nov., sp. nov., a bacterium isolated from the gorgonian octocoral Antillogorgia elisabethae, belonging to the family Pseudobacteriovoracaceae fam. nov., within the order Bdellovibrionales. Int J Syst Evol Microbiol. 2015 65: 522-30.
9. Duncan K.R., Haltli B., Gill K.A., Correa H., Berrué F., Kerr R.G. Exploring the diversity and metabolic potential of actinomycetes from temperate marine sediments from Newfoundland, Canada. J Ind Microbiol Biotechnol. 2015 42: 57-72.
10. Janso J.E., Haltli B.A., Eustáquio A.S., Kulowski K., Waldman A.J., Zha L., Nakamura H., Bernan V.S., He H., Carter G.T., Koehn F.E., Balskus E.P. Discovery of the lomaiviticin biosynthetic gene cluster in Salinispora pacifica. Tetrahedron. 2014 70: 4156-4164.
11. Duncan K., Haltli B., Gill K. A., Kerr R.G. Bioprospecting from marine sediments of New Brunswick, Canada: exploring the relationship between total bacterial diversity and Actinobacteria diversity. Mar Drugs. 2014 12: 899-925.
12. Correa H., Haltli B., Duque C., Kerr R.G. Bacterial communities of the gorgonian octocoral Pseudopterogorgia elisabethae Microb Ecol. 2013 66: 972-85.
13. Pike R.E., Haltli B., Kerr R. G. Description of Endozoicomonas euniceicola sp. nov. and Endozoicomonas gorgoniicola sp. nov., bacteria isolated from the octocorals Eunicea fusca and Plexaura sp., and an emended description of the genus Endozoicomona. Int J Syst Evol Microbiol. 2013 63: 4294-4302.
14. McCulloch M. W. B., Haltli B., Marchbank D. H., Kerr R. G. Evaluation of pseudopteroxazole and pseudopterosin derivatives against Mycobacterium tuberculosis and other pathogens. Mar Drugs. 2012 10: 1711-1728.
15. McCulloch M.W.B., Berrue F., Haltli B., Kerr R.G. One-pot syntheses of pseudopteroxazoles from pseudopterosins: a rapid route to non-natural congeners with improved antimicrobial activity. J Nat Prod. 2011 74: 2250-2256.
16. Jiang H., Haltli B., Feng X., Cai P., Summers M., Lotvin J., He M. Investigation of the biosynthesis of the pipecolate moiety of neuroprotective polyketide meridamycin. J Antibiot 2011 64: 533-538.
17. Berrue F., Withers S. T., Haltli B., Withers J., Kerr R.G. Chemical screening method for the rapid identification of microbial sources of marine invertebrate-associated metabolites. Mar Drugs. 2011 9: 369-381.
18. Liu H., Jiang H, Haltli B., Kulowski K., Muszynska E., Feng X., Summers M., Young M., Graziani E., Koehn F., Carter G. T., Min H. Rapid cloning and heterologous expression of the meridamycin biosynthetic gene cluster using a versatile Escherichia coli-Streptomyces artificial chromosome vector, pSBAC. J Nat Prod. 2009 72 (3): 389-395.
19. Ratnayake A. S., Haltli B., Feng X., Bernan V. S., Singh M. P., He H., Carter G.T. Investigating the biosynthetic origin of the nitro group in pyrrolomycins. J Nat Prod. 2008 71 : 1923–1926.
20. He M., Haltli B., Summers M., Feng X., Hucul J.A. Isolation and characterization of the meridamycin biosynthetic gene cluster from Streptomyces species NRRL 30748. Gene. 2006 377: 109-118.
21. Magarvey N. A., Haltli B., He M., Greenstein M. and Hucul J.A. Biosynthetic pathway for mannopeptimycins, lipoglycopeptide antibiotics active against drug-resistant gram-positive pathogens. Antimicrob Agents Chemother. 2006 50: 2167-2177.
22. Haltli B., Tan Y., Magarvey N.A., Wagenaar M., Yin X., Greenstein M., Hucul J.A., Zabriskie T.M. Investigating beta-hydroxyenduracididine formation in the biosynthesis of the mannopeptimycins. Chem Biol. 2005 12(11): 1163-1168.
23. Ritacco F. V., Haltli B., Janso J. E., Greenstein M., Bernan V.S. Dereplication of Streptomyces soil isolates and detection of specific biosynthetic genes using an automated Ribotyping instrument. J Ind Microbiol Biotechnol. 2003 30: 472-479.
Logan MacIntyre
Research Interests:
Hope Igboeli
 Research Interests:
Research Interests:Chris Cartmell
Research Interests:
I am a driven natural product chemist with a strong interest in mass spectrometry and the discovery and development of new bioactive natural products and their analogues from both marine and terrestrial sources. Completing my BSc at the University of Bangor in north Wales, an area of outstanding natural beauty, cultivated my interest for natural products which lead to my PhD studies at the University of St Andrews under the supervision of Professor Rebecca Goss. My current research focuses on identification, isolation, and structural elucidation of novel natural products along with the development of accessing silent or cryptic biosynthetic pathways.
Education:
2015 - 2019 - PhD Chemistry, University of St Andrews, Scotland, United Kindgom
2012 - 2015 - BSc Chemistry, University of Bangor, Wales, United Kingdom
Publications:
M. M. LeClair, Z. Maw, A Grunwald, J. R. Kelly, R.G. Kerr and C. Cartmell*. The Levesquamides: A Family of Isothiazoline Natural Products. Submitted to Marine Drugs 2022
C. Cartmell, A. L. Grunwald, C. Arango, N. J. Duncan, B. A. Haltli, L. E. Diaz and R. G. Kerr, Draft Genome Sequence of Streptomyces sp. RKCA744 Isolated from the Arauca River, Colombia. Microbial Resource Announcements. 2022
E. Pope, C. Cartmell, B. A. Haltli, R.G. Kerr and A. Ahmadi. Microencapsulation and In-SituIncubation to Improve the Recovery of Bacteria from Marine Habitats. Frontiers in Microbiology 2022
C. Cartmell, S. K. Wheatly, B. A. Haltli, R.G. Kerr and A. Ahmadi. Development of a Micro-Microbe Domestication Pod for In-Situ Cultivation. RSC advances 2022
Z. S. S. Al-Taie, C. Cartmell, R. T. Froom, R. G. Kerr, P. J. Murphy* and A. van Teijlingen. Synthesis of (+)-(R)-Tiruchanduramine. Molecules 2022
E. Naseri, C. Cartmell, M. Saab, R. G. Kerr and A. Ahmadi. Development of NOCC-Starch Biomaterial Inks for 3D Printed Wound Dressing Applications. Macromolecular Biosciences. 2021
A. L. Grunwald, C. Cartmell, R. G. Kerr. Auyuittuqamides A-D, Cyclic Decapeptides from Sesquicillium microsporum RKAG 186 Isolated from Frobisher Bay. Journal of Natural Products. 2020
T. Alkayyali, E. Pope, S. K. Wheatley, C. Cartmell, B. A. Haltli, Russ G. Kerr, and A. Ahmadi. Development of Microbe Domestication Pod (MD Pod) for In-Situ Cultivation of Micro-encapsulated Marine Bacteria. Biotechnology and Bioengineering. 2020
C. Cartmell, A. Abou Fayad, R. Lynch, S. V. Sharma, N. Hauck, B. Gust, R. J. M. Goss. SynBio-SynChem Approaches to Diversifying the Pacidamycins through the Exploitation of an Observed Pictet- Spengler Reaction. ChembioChem, 2020
E. Naseri, C. Cartmell, M. Saab, R. G. Kerr and A. Ahmadi. Development of 3D printed drug-eluting scaffolds for preventing piercing infection. Pharmaceutics, 12, (9), 901, 2020
C. Cartmell, C. Pubill-Ulldemolins, S. V. Sharma, J. Zhao, P. Cárdenas, R. J. M. Goss. Diversification Through Heck Cross-Coupling of Indole-Based Substrates in H2O: From Unprotected Halotryptophans to Halotryptophans in a Natural and a New to Nature Natural Product. Chemistry, a European journal. (25) 2019
Y. Renault, R. Lynch, E. Marelli, S. V. Sharma, J. Sharp, C. Cartmell, P. Cárdenas, R. J. M. Goss. Buchwald Hartwig Diversification of Unprotected Halotryptophans, Halotryptophan Containing Tripeptides and the Natural Product Barettin in Aqueous Conditions. Chemical communications. 2019
C. Cartmell, D. M. Evans, J. M. L. Elwood, H. S. Fituri, P. J. Murphy T. Caspari, B. Poniedziałek, P. Rzymski. Synthetic Analogues of Cyanobacterial Alkaloid Cylindrospermopsin and Their Toxicological Activity. Toxicology In Vitro. (44): 172-181. 2017
M. J. Corr, S. V. Sharma, C. Pubill-Ulldemolins, R. T. Bown, P. Poirot, D. R. M. Smith, C. Cartmell, A. Abou Fayad, R. J. M. Goss. Sonogashira Diversification of Unprotected Halotryptophans, Halotryptophans Containing Tripeptides and Generation of a New to Nature Bromo-Natural Product and its Diversification in Water. Chemical Science. (8): 2039-2046. 2017
S. V. Sharma, X. Tong, C. Pubill-Ulldemolins, C. Cartmell, E. J. A. Bogosyan, E. J. Rackham, E. Marelli, R. B. Hamed, R. J. M. Goss. Living GenoChemetics by Hyphenating Synthetic Biology and Synthetic Chemistry In Vivo. Nature Communications. (8) 2017
Contact:
Email: ccartmell@upei.ca
Phone: 1-902-620-5035
Doug Marchbank
 Research Interests:
Research Interests:Nautilus colleagues
Alumni
|
Former Lab Members |
|||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 Kelly Richard
Research assistant
|
 Jeanna McLeod
BSc 2011
|
||||||||||||||
|
Saskia Hart
Research Assistant
|
Katherine Duncan
PhD 2012
|
 Katheryn Hay
BSc 2012
|
|||||||||||||
 Christie Ings
BSc 2012
|
 Jeanetta McGinley
BSc 2012
|
 David Forner
BSc 2012
|
 Troy Cumberlach
Visiting Student 2011-2012
|
||||||||||||
 Ann Hanseebaran
Visiting Student 2011-2012
|
 Andi Hanif
Post Doc 2011-2012
|
 Quinn Gill
Summer Student 2013
|
|||||||||||||
Stacey Goldberg
Research Interests:
Goldberg S., Mueller S., Brichetti J., Sadoff J. Cryo-preserved Whole Blood (WB) for use in Cell Mediated Immunity (CMI) Assays, Poster on behalf of Aeras Global TB Vaccine Foundation. TB Vaccines for the World Conference, Atlanta, GA (2008).
Casey, J.R., Aucan J.P., Goldberg S.R., Lomas M.W. Changes in partitioning of carbon amongst autotrophic pico- and nanoplankton groups in response to changes in the North Atlantic Oscillation. Deep Sea Research II: Topical Studies in Oceanography (Feb 2013).
Goldberg, S. Advancement of molecular methods for identification of bromotyrosine alkaloid derivatives from marine sponges and assessment of their therapeutic potential. Graduate Studies and Research Days, AVC, University of Prince Edward Island (2013). Gold Prize Award.
Leon Liang
 Research Interests:
Research Interests:2011-2013 Chemical Engineering, Hainan University
2013-present (transfer) Chemistry(Undergraduate), UPEI
Group Activities
|
Fun Events Outside the Lab
|
|
 |
Kerr Lab Christmas 2014 The lab spent the evening celebrating the holiday season with good food and drink and a gift exchange. |
 |
Kerr curling outing The lab spent the afternoon at the rink on April 1st 2012 |
 |
1st Kerr Golf Tournament The lab spend the afternoon at the green for the first annual Kerr Golf Tournament on June 24th 2011 |
|
|
Deep Sea Fishing Adventure The lab tried out their fishing techniques at Wade's Deep Sea Fishing in September 2010 |
 |
Maritime Natural Products Conference 2010 The second annual Maritime Natural Products Conference was held at UPEI August 31 - September 2, 2010 |
 |
Lab Retreat 2010 First Annual Kerr Group Lab Retreat at Camp Riverdale July 2010 |
 |
Football at Greenwich National Park
Our football skills were put to the test on this blustery day in late May, 2009. The boardwalk was beautiful, the scenery was stunning, the air was fresh, and the food at PEI's best "Fish & Chips" spot was delicious!
|
.jpg) |
Skating on the "Puddle"
Every now and again the unversity campus acquires its own pond, which is really nothing more than a huge puddle. This November, we decided to test our skating skills -- for some of us it was the first time in years! It was a lunch break to remember!
|
 |
Skiing at Brookvale
Our lab members come from many different countries. Some of them saw snow for the first time this winter! We decided to give them a true PEI welcome at Brookvale Provincial Ski Park.
|
 |
IUPAC Conference
On July 13th to 18th, 2008, we hosted the IUPAC Conference on Biodiverstiy and Natural Products. The program included plenary lectures, invited lectures, and Oral/Poster presentations, as well as plenty of opportunities to tour our beautiful island! The conference finale included a catered lobster meal on a beach.
|
 |
Bioquest
On October 17, 2008, the Kerr group participated in Bioquest, during which 200 grade five students from around the Maritimes are invited to come and enjoy hands-on demonstrations of science in the real world. We had a fantastic day with these young and enthusiastic learners.
|
.jpg) |
Party at the Cottage!
We kicked off the summer of 2008 in style at Russ and Stacey's cottage. There was badminton, soccer, swimming, boating,clam-eating, and of course, a delicious barbeque on the deck!
|
IUPAC 2008
.jpg) |
 |
 |
 |
 |
 |
 |
 |
.jpg) |
 |
 |
 |
Kerr Lab Christmas 2014
 |
|
Party at the Cottage
 |
 |
 |
.jpg) |
.jpg) |
.jpg) |
Skating on the "Puddle"
.jpg) |
|
.jpg) |
 |
 |
 |
 |
 |
Skiing at Brookvale
 |
|
.jpg) |
 |
 |
 |
Bioquest
 |
 |
 |
 |
 |
 |
 |
 |
Football at Greenwich
.jpg) |
 |
 |
 |
 |
 |
 |
 |
 |
 |
Field Work

San Salvador 2014
 |
Examining samples in the wet lab |
 |
Gorgonian growth rate study. |
 |
Demonstration of water pumping action of a sponge. |
 |
Turkey 2012
 |
The town of Kas, Turkey – the departure location of one of our dive trips. |
 |
Brad – collecting in the murky waters. |
 |
Bulent (our local guide and sponge taxonomist) with Brad on hotel balcony (= temporary lab). |
 |
Brad processing samples in hotel room. |
San Salvador 2012
 |
The group visiting the Columbus monument. |
 |
A picnic in the back of one of the Gerace trucks! |
 |
Setting up a gorgonian Aquaculture project. |
2011 Expedition to Iqaluit, Nunavut
Exploring Canada's Arctic as a Source of Novel Natural Products
Brad Haltli, September 2011
Canada's arctic is a cold and forbidding region of the world. This diverse area is often thought to be relatively devoid of biolgical diversity and most people associate the arctic with relatively few large mammals such as polar bears, muskox, seals, walruses and whales, as well as a variety of migratory bird species. This view of the arctic largely ignores microbial diversity. Several studies in the arctic have examined general bacterial diversity of tundra and the diversity observed has rivalled that of tropical soils. The majority of the studies conducted to date have been concerned with bacterial diveristy as it relates to climate change and bioremediation of contaminated soils. The aim of the 2011 expedition to Iqaluit was to obtain a number of terrestrial soil and intertidal marine sediment samples so that we could assess microbial diversity of this arctic region from a natural product discovery perspective. We will characterize the microbial diversity using a combination of culture independent and culture dependent methodologies. We will subsequently evaluate bacteria cultured from these samples for the ability to produce novel bioactive metabolites. Our hypothesis is that novel bacterial and fungal biodiversity will be discovered from arctic samples which will coincided with the production of novel natural products from these unique microbes.
Below are some photographs that document this expedition:
























 .
.



2010 Expedition to Barbados
|
Februrary 2010 Trip to Barbados Grad students Jennifer Cuillerier, Rebecca Pike, and Beth Pearce accompanied Brad Haltli, and Russ Kerr for a week in Barbados to conduct field work. We were joined by two graduate students in Dr. Tinto’s group – Ann Seebaran and Troy Cumberbatch. Collections of invertebrates were conducted for the purpose of isolating microbes for drug discovery projects, and Jenn, Becca and Beth conducted field work for a Marine Natural Products course. |
 |
 Beach across the road from rental house
|
 Becca, Russ, Jenn and Winston
|
 Becca, Beth and Jenn
|
 Ann, Troy and Jenn outside the dive shop Ann, Troy and Jenn outside the dive shop |
 Becca and Jenn getting ready for a dive
|
 Jenn, Russ, Ann, Troy, Brad, Becca and Beth
|
 |
 |
 |
 Renatta Goodridge, local sponge
taxonomist (UWI) joined us for a few dives.
|
 |
 |
 |
 |
 One of the colourful dive boats that we used.
|
|
2009 Expedition to Barbados
|
November 2009 Collection Trip to Barbados Fabrice Berrue, David Overy and Russ Kerr traveled to Barbados to initiate a collaboration with Dr. Winston Tinto (University of West Indies, Cave Hill). Collections were focused on the south coast of the island using the services of EcoDivers. The goals of the trip were to collect invertebrates and algae and also to isolate microbes from selected invertebrate samples. |
 |
 |
 Dr. Motra-Meira’s lab (UWI) – graciously made available to us for sample processing and microbiology work
|
 Fabrice – ready to dive!
|
 Fabrice and David collecting
|
2009 Expedition to Newfoundland
.
|
Newfoundland August 2009 During the summer of 2009 we enjoyed various collecting trips to regions of PEI, the Bay of Fundy and Newfoundland. |
|
 |
 |
 |
 |
 Ian and Gavin – our young helpers for the week
|
 Lots of great lab space at Bonne Bay
|
2008 Expedition to the Bahamas
|
Our 2008 Collection Trip to the Bahamas
|
|
|
|
In August, we visited the Gerace Research Centre located on San Salvador in the Commonwealth of the Bahamas. This was a highly successful trip with samples collected from a variety of habitats including reefs, caves and marine lakes. Samples are now being processed for our drug discovery program and microbiological studies.
|
 |
|
 An aereal view of the airport on San Salvador - one of our dive sites is directly off shore from the runway.
|
 The Gerace cafeteria.
|
|
 Our dive boat for the week.
|
 Dovi, Veronica, Fabrice, Doug and Russ
|
|
 Stacey collecting
|
 Off on the search...
|
|
 |
 Stacey meets a very friendly grouper known by the locals as Sponge Bob!
|
|
 A specimen of Pseudopterogorgia elisabethae in the ziploc bag ready for micro work back at the lab.
|
 Collecting a gorgonian for microbiological work.
|
|
 Brad and Fabrice collecting Erythropodium.
|
 Brad - off to the lab.
|
|
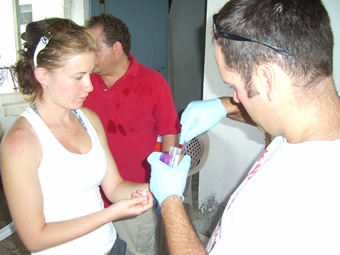 Veronica and Brad processing samples for microbiological work.
|
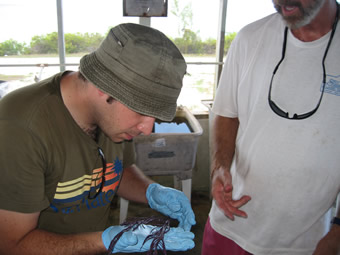 Fabrice examining a gorgonian. Fabrice examining a gorgonian. |
|
|
|
|
|
|
|
|
|
|
|
||
2007 Expedition to the Florida Keys
| The trip to Florida was largely directed at collections of invertebrates and algae for a drug discovery program. Rather than staying on a research vessel as we have done in the past, this trip involved small boats with hotel rooms sometimes being used as a lab! Collections were made off Boca Raton, Key Largo, Marathon Key and Summerland Key. At Summerland Key, we stayed at the Mote Tropical Research Lab which is an ideal location for such field work | |
 |
 |
 |
 |
Publications
120. Sperlich J, Kerr R, Teusch N. The Marine Natural Product Pseudopterosin Blocks Cytokine Release of Triple-Negative Breast Cancer and Monocytic Leukemia Cells by Inhibiting NF-κB Signaling. Mar Drugs. 2017 Aug 23;15(9). pii: E262. doi: 10.3390/md15090262. PubMed PMID: 28832545; PubMed Central PMCID: PMC5618401.
119. Overy D, Correa H, Roullier C, Chi WC, Pang KL, Rateb M, Ebel R, Shang Z, Capon R, Bills G, Kerr R. Does Osmotic Stress Affect Natural Product Expression in Fungi? Mar Drugs. 2017 Aug 13;15(8). pii: E254. doi: 10.3390/md15080254. PubMed PMID: 28805714; PubMed Central PMCID: PMC5577608.
118. Grunwald AL, Berrue F, Robertson AW, Overy DP, Kerr RG. Mortiamides A-D, Cyclic Heptapeptides from a Novel Mortierella sp. Obtained from Frobisher Bay. J Nat Prod. 2017 Oct 27;80(10):2677-2683. doi: 10.1021/acs.jnatprod.7b00383. Epub 2017 Sep 18. PubMed PMID: 28921982.
117. Forget SM, Robertson AW, Overy DP, Kerr RG, Jakeman DL. Furan and Lactam Jadomycin Biosynthetic Congeners Isolated from Streptomyces venezuelae ISP5230 Cultured with N(ε)-Trifluoroacetyl-l-lysine. J Nat Prod. 2017 Jun 23;80(6):1860-1866. doi: 10.1021/acs.jnatprod.7b00152. Epub 2017 May 18. PubMed PMID: 28520425.
116. Abd-El-Aziz AS, Agatemor C, Etkin N, Bissessur R, Overy D, Lanteigne M, McQuillan K, Kerr RG. Quaternized and Thiazole-Functionalized Free Radical-Generating Organometallic Dendrimers as Antimicrobial Platform against Multidrug-Resistant Microorganisms. Macromol Biosci. 2017 Jul;17(7). doi:10.1002/mabi.201700020. Epub 2017 Mar 31. PubMed PMID: 28371348.
115. Robertson V, Haltli B, McCauley EP, Overy DP, Kerr RG. Highly Variable Bacterial Communities Associated with the Octocoral Antillogorgia elisabethae. Microorganisms. 2016 Jul 5;4(3). pii: E23. doi: 10.3390/microorganisms4030023. PubMed PMID: 27681917; PubMed Central PMCID: PMC5039583.
114. Tangerina MM, Correa H, Haltli B, Vilegas W, Kerr RG. Bioprospecting from cultivable bacterial communities of marine sediment and invertebrates from the underexplored Ubatuba region of Brazil. Arch Microbiol. 2017 Jan;199(1):155-169. doi: 10.1007/s00203-016-1290-9. Epub 2016 Sep 19. PubMed PMID: 27644133.
113. McCauley EP, Haltli B, Correa H, Kerr RG. Spatial and temporal investigation of the microbiome of the Caribbean octocoral Erythropodium caribaeorum. FEMS Microbiol Ecol. 2016 Sep;92(9). pii: fiw147. doi: 10.1093/femsec/fiw147. Epub 2016 Jul 4. PubMed PMID: 27381833.
112. Sommer B, Overy DP, Haltli B, Kerr RG. Secreted lipases from Malassezia globosa: recombinant expression and determination of their substrate specificities. Microbiology. 2016 Jul;162(7):1069-79. doi: 10.1099/mic.0.000299. Epub 2016 Apr 29. PubMed PMID: 27130210.
111. Herzog B, Overy DP, Haltli B, Kerr RG. Discovery of keratinases using bacteria isolated from marine environments. Syst Appl Microbiol. 2016 Feb;39(1):49-57. doi: 10.1016/j.syapm.2015.10.004. Epub 2015 Nov 9. PubMed PMID: 26607323.
110. Abd-El-Aziz AS, Agatemor C, Etkin N, Overy DP, Lanteigne M, McQuillan K, Kerr RG. Antimicrobial Organometallic Dendrimers with Tunable Activity against Multidrug-Resistant Bacteria. Biomacromolecules. 2015 Nov 9;16(11):3694-703. doi: 10.1021/acs.biomac.5b01207. Epub 2015 Oct 16. PubMed PMID: 26452022.
109. Johnston CW, Connaty AD, Skinnider MA, Li Y, Grunwald A, Wyatt MA, Kerr RG, Magarvey NA. Informatic search strategies to discover analogues and variants of natural product archetypes. J Ind Microbiol Biotechnol. 2016 Mar;43(2-3):293-8. doi: 10.1007/s10295-015-1675-9. Epub 2015 Sep 8. PubMed PMID: 26350080.
108. Sommer B, Overy DP, Kerr RG. Identification and characterization of lipases from Malassezia restricta, a causative agent of dandruff. FEMS Yeast Res. 2015 Nov;15(7). pii: fov078. doi: 10.1093/femsyr/fov078. Epub 2015 Aug 21. PubMed PMID: 26298017.
107. Poddutoori PK, Bregles LP, Lim GN, Boland P, Kerr RG, D'Souza F. Modulation of Energy Transfer into Sequential Electron Transfer upon Axial Coordination of Tetrathiafulvalene in an Aluminum(III) Porphyrin-Free-Base Porphyrin Dyad. Inorg Chem. 2015 Sep 8;54(17):8482-94. doi: 10.1021/acs.inorgchem.5b01190. Epub 2015 Aug 13. PubMed PMID: 26270270.
106. Arens JC, Haltli B, Kerr RG. Draft Genome Sequence of Kitasatospora griseola Strain MF730-N6, a Bafilomycin, Terpentecin, and Satosporin Producer. Genome Announc. 2015 Mar 26;3(2). pii: e00208-15. doi: 10.1128/genomeA.00208-15. PubMed PMID: 25814608; PubMed Central PMCID: PMC4384148.
105. Gill KA, Berrué F, Arens JC, Carr G, Kerr RG. Cystargolides, 20S Proteasome Inhibitors Isolated from Kitasatospora cystarginea. J Nat Prod. 2015 Apr 24;78(4):822-6. doi: 10.1021/np501060k. Epub 2015 Mar 13. PubMed PMID: 25769015.
104. McCauley EP, Haltli B, Kerr RG. Description of Pseudobacteriovorax antillogorgiicola gen. nov., sp. nov., a bacterium isolated from the gorgonian octocoral Antillogorgia elisabethae, belonging to the family Pseudobacteriovoracaceae fam. nov., within the order Bdellovibrionales. Int J Syst Evol Microbiol. 2015 Feb;65(Pt 2):522-30.
103. David Overy, Paul Bayman, Russell Kerr and Gerald Bills. An assessment of natural product discovery from marine (sensu strictu) and marine-derived fungi. Mycology: an International Journal on Fungal Biology, 5, 145-167 (2014).
102. David P. Overy, Fabrice Berrue, Hebelin Correa, Novriyandi Hanif, Kathryn Hay, Martin Lanteigne, Kathrine McQuilian, Stephanie Duffy, Patricia Boland, Ramesh Jagannathan, Gavin S. Carr, Marieke Vansteeland & Russell G. Kerr. Sea foam as a source of fungal inoculum for the isolation of biologically active natural products. Mycology: an International Journal on Fungal Biology. DOI:10.1080/21501203.2014.931893 (2014).
101. Krista A. Gill, Fabrice Berrué, Jennifer C. Arens and Russell G. Kerr. Isolation and structure elucidation of cystargamide, a lipopeptide from Kitasatospora cystarginea. J Nat Prod DOI 10.1021/np500122s (2014).
100. Michelle A. Markus, Jonathan Ferrier, Sarah M. Luchsinger, Jimmy Yuk, Alain Cuerrier, Michael J. Balick, Joshua M. Hicks, K. Brian Killday, Christopher W. Kirby, Fabrice Berrue, Russell G. Kerr, Kevin Knagge, Tanja Gödecke, Benjamin E. Ramirez, David C. Lankin, Guido F. Pauli, Ian Burton, Tobias K. Karakach, John T. Arnason, Kimberly L. Colson. Distinguishing Vaccinium Species By Chemical Fingerprinting Based on NMR Spectra, Validated with Spectra Collected in Different Laboratories" Planta Medica 80, 732-739 (2014).
99. Michael R. van den Heuvel, Natacha S. Hogan, Gillian Z. MacDonald, Fabrice Berrue, Rozlyn F. Young, Collin J. Arens, Russell G. Kerr, Phillip M. Fedorak. Assessing accumulation and biliary excretion of naphthenic acids in yellow perch exposed to oil sands-affected waters. Chemosphere 95, 619–627 (2014).
98. E. Marchal, Md. Uddin, D. SMithen, C. Hawco, M. Lanteigne, D. Overy, R. Kerr and A. Thompson. Antimicrobial activity of non-natural prodigiosenes. RSC Advances, 3, 22967-22971 (2013).
97. Gavin Carr, Fabrice Berrue, Saranyoo Klaiklay, Isabelle Pelletier, Russell G. Kerr. Natural products with protein tyrosine phosphatase inhibitory activity. Methods, 65, 229-238 (2013).
96. Rebecca E. Pike, Brad Haltli, and Russell G. Kerr. Description of Endozoicomonas eunicicola sp. nov. and Endozoicomonas gorgonicola sp. nov., Bacteria Specifically Associated with the Gorgonian Octocorals, Eunicea fusca and Plexaura sp., and an emended description of the genus Endozoicomonas. Int. Journal of Systematic and Evolutionary Microbiology 63, 4294 - 4302 (2013).
95. David Forner, Fabrice Berrué, Hebelin Correa, Katherine Duncan, and Russell G Kerr. Chemical dereplication of marine actinomycetes by LC-HRMS profiling and statistical analysis. Analytica Chemica Acta, 805, 70 - 79 (2013).
94. Jennifer Arens, Fabrice Berrue, Jason Pearson and Russell Kerr. Isolation and structure elucidation of satosporin A and B: new polyketides from Kitasatospora griseola. Organic Letters, 15, 3864 - 3867 (2013).
93. Hebelin Correa, Brad Haltli, Carmenza Duque, Russell Kerr. Bacterial Community of the Gorgonian Octocoral Pseudopterogorgia elisabethae. Microbial Ecology 66, 972 – 985 (2013).
92. Ghada Ebead, David Overy, Russell Kerr. Westerdykella reniformis sp. nov., producing the antibiotic metabolites melinacidin IV and chetracin B. IMA Fungus, 189-201 (2012).
91. Fabrice Berrue, Malcolm McCulloch, Patricia Boland, Saskia Hart, Mary Kay Harper, James Johnson, Russell G. Kerr. Isolation of steroidal glycosides from the Caribbean sponge Pandaros acanthifolium. J. Nat. Prod. 75, 2094- (2012).
90. Angela Duque-Alarcon, Lory Santiago-Vazque, Russell Kerr. Microbial community analysis of the octocoral Eunicea fusca. Electronic Journal of Biotechnology, Vol. 15, no. 5 (2012).
89. Malcolm McCulloch, Brad Haltli, Douglas Marchbank, Russell Kerr. Evaluation of pseudopteroxazole and pseudopterosin derivatives against Mycobacterium tuberculosis and other pathogens. Marine Drugs, 11, 1711 – 1728 (2012).
88. Douglas H. Marchbank, Fabrice Berrue, Russell Kerr. Eunicidiol, a new anti-inflammatory dilophol diterpene from Eunicea fusca. J. Nat. Prod. 75 (7), 1289-93 (2012).
87. Hebelin Correa, Fabrice Berrué, Brad Haltli, Carmenza Duque, Russell Kerr. Rapid construction of a library of natural products from the cultivation of 14 bacteria of the phylum Firmicutes, symbionts of the octocoral Pseudopterogorgia elisabethae of Isla de Providencia. J. Colombian Academy of Science (Revista de la Academia Colombiana de Ciencias), 35(136), 337-348 (2011).
86. Malcolm McCulloch, Fabrice Berrue, Brad Haltli and Russell Kerr. One-pot syntheses of pseudopteroxazoles: a rapid route to non-natural congeners with improved antimicrobial activity. J. Nat. Prod. 74, 2250-2256 (2011).
85. Fabrice Berrue, Malcolm McCulloch, Russell Kerr. Marine diterpene glycosides. Bioorganic & Medicinal Chemistry 19, 6702-6719 (2011).
84. Fabrice Berrue, Sydnor T. Withers, Brad Haltli, Jo Withers, and Russell G. Kerr. Chemical screening method for invertebrate-associated bacteria involved in the production of secondary metabolites present in the host organism. Marine Drugs, 9, 369-381 (2011).
83. Douglas H. Marchbank and Russell G. Kerr. Semisynthesis of fuscoside B analogues and eunicosides, and analysis of anti-inflammatory activity. Tetrahedron, 67, 3053-3061 (2011).
82. Hebelin Correa, Fabio Aristizabal, Carmenza Duque and Russell Kerr. Cytotoxic and Antimicrobial Activity of Pseudopterosins and seco-Pseudopterosins Isolated from the Octocoral Pseudopterogorgia elisabethae of San Andrés and Providencia Islands (SW Caribbean). Marine Drugs, 9, 334-344 (2011).
81. Jose Lopez, A. Ledger, Lory Santiago-Vázquez, M. Pop, Llanie Ranzer, Richard Feldman, Russell G. Kerr. Suppression subtractive hybridization PCR isolation of cDNAs from a Caribbean soft coral. Electronic Journal of Biotechnology, 1418 01 (2011).
80. Maysoon Saleh and Russell G. Kerr. Identification of the cyclase product and its first oxidation product in the biosynthesis of fuscol and fuscosides. Aust. J. Chem. 63, 901-906 (2010). Invited contribution.
79. Abhijeet S. Kate, Amber C. Kohl and Russell G. Kerr. A simple and sensitive APCI-LC-MS method for the detection of the antitumor agent, carmustine (BCNU) in rat plasma. J. Liq. Chromatogr. & Related Technol. 33, 818-824 (2010).
78. Abhijeet S. Kate, Kelly Richard, Balaji Ramanathan, and Russell G. Kerr. A halogenated pseudopterane diterpene from the Bahamian octocoral Pseudopterogorgia acerosa. Can. J. Chem. 88, 318-322 (2010).
77. Jose Lopez, Benoit Mouzon, Peter McCarthy, Russell Kerr. The many faces of gene expression profiling: transcriptome analyses applied towards elucidating marine organismal interactions and metabolism. Textbook on Molecular Biotechnology. IK International Publishing House Pvt. Ltd (AUUP), ISBN 978-93-80026-37-4. Pp 287-304 (2009).
76. Abhijeet Kate, Jason Pearson, Balaji Ramanathan, Kelly Richard, Russell Kerr. Isolation, biomimetic synthesis and biological activity of bis-pseudopterane amines. J. Nat. Prod. 72(7), 1331-1334 (2009).
75. Fabrice Berrue, Abdelnasser Ibrahim, Patricia Boland and Russell Kerr. A newly isolated marine Bacillus pumilus SP21: a source of novel lipoamides and other antimicrobial agents. Pure and Applied Chemistry 81, 1027-1031 (2009).
74. Jamie Frenz and Russell Kerr. Sesquiterpene variability in the Gorgonian Genus Plexaurella. Comp. Biochem. and Physiol. C 150, 125-131 (2009)
73. Jose Lopez, Lanie Ranzer, A. Ledger, Schoch, Alan Duckworth, Peter McCarthy and Russell Kerr. Comparison of Bacterial Diversity within the Coral Reef Sponge, Axinella corrugata and the Encrusting Coral Erythropodium caribaeorum. Proceedings of the 11th International Coral Reef Symposium. Florida, 1355 - 1359, (2009)
72. Russell Kerr. Pseudopterosin production: the promise and challenges. Chapter in “Vision and Future of Investigations in Parasitology and Natural Products” Republic of Columbia, Bogota, pgs 399 – 410, (2009).
71. Fabrice Berrue and Russell G. Kerr. Dipterpenes from gorgonian corals. Nat. Prod. Rep., 26, 681–710 (2009).
70. Llanie K Ranzer, Thomas Brueck, Wolfram Brueck, Jose Lopez, Russell Kerr. A new prokaryotic farnesyldiphosphate synthase from the octocoral Eunicea fusca: differential display, inverse PCR, cloning and characterization. Marine Biotechnology, 11, 62 (2009).
69. Abhijeet Kate, Isabelle Aubry, Michel Tremblay and Russell Kerr. Lipidyl pseudopteranes A-F: isolation, biomimetic synthesis and PTP1B inhibitory activity of a novel class of pseudopteranoids from the gorgonian Pseudopterogrogia acerosa. J. Nat. Prod., 71, 1977-1982 (2008).
68. Angela P. Duque-Alarcón, Lory Z. Santiago-Vázquez, Julie Enticknap, Nasser Alam and Russell Kerr Diversity of Coral Eunicea fusca Associated Bacteria Using Culture Dependent Techniques. Res. J. Microbiol. 10: 614-621, (2008).
67. Mohammad Al-Ghoul, Thomas Brueck, Janelle Lauer-Fields, Victor Asirvatham, Claudia Zapata, Russell Kerr and Gregg Fields. Comparative proteomic analysis of matched primary and metastatic melanoma cell lines. Journal of Proteome Research, 7 (9) 4107–4118, (2008).
66. Jamie L. Frenz-Ross, Julie J. Enticknap and Russell G. Kerr. The effect of bleaching on the terpene chemistry of Plexaurella fusifera: evidence that zooxanthellae are not responsible for sesquiterpene production. Marine Biotechnology, 10, 572-578 (2008).
65. Lory Z. Santiago-Vázquez, Thomas B. Brück, Wolfram M. Brück, Angela P. Duque-Alarcón, Peter J. McCarthy and Russell G. Kerr. The diversity of the bacterial communities associated with the azooxanthellate hexacoral Cirrhipahtes lutkeni. Journal of the International Society for Microbial Ecology, 1, 654-659 (2007).
64. Lory Z. Santiago-Vázquez, Nealie C. Newberger, and Russell G. Kerr. Cryopreservation of the dinoflagellate symbiont of the octocoral Pseudopterogorgia elisabethae. Marine Biology, 152, 549-556 (2007).
63. Thomas Brueck, Wolfram Brueck, Lory Z. Santiago-Vázquez, Peter McCarthy and Russell G. Kerr. Diversity of the bacterial communities associated with the azooxanthellate deep water octocorals Leptogorgia minimata, Iciligorgia schrammi and Swiftia exertia. Marine Biotechnology, 9, (2007).
62. Xing Dai, Zhongliang Wan, Russell G. Kerr, and Huw M. L. Davies. Synthetic and isolation studies related to the marine natural products (+)-elisabethadione and (+)-elisabethamine. J. Org. Chem. 72, 1895-2000 (2007).
61. Lory Z. Santiago-Vázquez, Llanie K. Ranzer, and Russell G. Kerr. Comparison of two total RNA extraction protocols using the marine gorgonian coral Pseudopterogorgia elisabethae and its symbiont Symbiodinium sp. Electronic Journal of Biotechnology, 9(5), (2006).
60. M. Isabel Nieto, Noemi Gonzalez, Jamie Rodriguez, Russell G. Kerr and Carlos Jimenez. New cytotoxic cembranolides: isolation, biogenetic studies and synthesis of analogues. Tetrahedron 62, 11747-11754 (2006).
59. Nealie Newberger, Llanie Ranzer, Jennifer Boehnlein and Russell Kerr. Induction of terpene biosynthesis in the dinoflagellate symbionts of the Caribbean gorgonian corals of the genera Pseudopterogorgia and Eunicea. Phytochem. 67, 2133-2139 (2006).
58. Russell G. Kerr, Amber Kohl and Tyrone Ferns. Elucidation of the biosynthetic origin of the anti-inflammatory pseudopterosins. J. Ind. Microbiol. Biotechnol. 33, 532-538 (2006).
57. Thomas Brueck and Russell Kerr. Purification and kinetic properties of elisabethatriene synthase from the coral Pseudopterogorgia elisabethae. Comp. Biochem. and Physiol. Part B 143, 269-278 (2006).
56. Tyrone Ferns and Russell Kerr. Oxidations of erogorgiaene in pseudopterosin biosynthesis. Tetrahedron 61, 12358-12365 (2005).
55. Jennifer Boehnlein, Lory Santiago-Vazquez and Russell Kerr. Diterpene biosynthesis by the dionflagellate symbiont of the Caribbean gorgognian Pseudopterogorgia bipinnata. Mar. Ecol. Prog. Ser. 303:105-111 (2005).
54. Tyrone Ferns and Russell Kerr. Identification of amphilectosins as key intermediates in pseudopterosin biosynthesis. J. Org. Chem. 70, 6152-6157 (2005).
53. Russell G. Kerr, Jennifer Boehnlein, Nealie Newberger and Llanie Ranzer. Concentrations of anti-inflammatory compounds increased in culture studies of coral, microalgae. Global Aquaculture Advocate, 70, (2004).
52. Jamie Frenz, Amber Kohl and Russell Kerr Kerr; Marine Natural Products as Therapeutic Agents – Part 2. Expert Opinion on Therpeutic Patents; 14, pp 17-33 (2004).
51. Amber Kohl and Russell Kerr. Identification and characterization of the pseudopterosin diterpene cyclase, elisabethatriene synthase, from the marine gorgonian, Pseudopterogorgia elisabethae. Arch. Biochem. Biophys. 424, 97-104 (2004).
50. Maysoon B. Saleh and Russell G. Kerr. Oxidation of Tyrosine Diketopiperazine to DOPA Diketopiperazine with Tyrosine Hydroxylase. J. Nat. Prod. 67, 1390-1391 (2004).
49. Amber Kohl and Russell Kerr. Pseudopterosin biosynthesis: Aromatization of the diterpene cyclase product, elisabethatriene. Marine Drugs, 1, 54-65 (2003).
48. Laura Mydlarz, Robert Jacobs, Jennifer Boehnlein and Russell Kerr. Evidence that the origin of pseudopterosinn biosynthesis resides in the dinoflagellate symbiont of Pseudopterogorgia elisabethae. Chemistry and Biology, 10, 1051-1056 (2003).
47. Amber Kohl and Russell Kerr A postulated biosynthetic origin of pseudopterosins and a proposed chemoenzymatic production method. J. Indust. Microbiol. 30, 495-499 (2003).
46. Athar Ata, Russell Kerr, Claudia Moya and Robert Jacobs. Identification of anti-inflammatory diterpenes from the marine gorgonian Pseudopterogorgia elisabethae. Tetrahedron. 59, 11389 (2003).
45. J. Cho, J. Choi, I Kong, S. Park, R. Kerr and Y. Hong. A procedure for axenic isolation of the marine microalgae Isochrysis galbana from heavily contaminated mass cultures. J. Appl. Phycol. 14, 385-390 (2002).
44. Noemí González, Jaime Rodríguez, Russell G. Kerr and Carlos Jiménez. Cyclobutenbriarein A, the First Diterpene with a Tricyclo[8.4.0.03,6]tetradec-4-ene Ring System from the Gorgonian Briareum asbestinium. J. Org. Chem. 67, 5117-5123, (2002).
43. Renee S. Thornton and Russell G. Kerr. Induction of Pseudopterosin Biosynthesis in the Gorgonian Pseudopterogorgia elisabethae. J. Chem. Ecol. 28, 2083-2090 (2002).
42. Jinghai Wen and Russell Kerr. Purification and Characterization of the Fatty Acid Synthase from Bugula neritina. Comp. Biochem. and Physiol. Part B, 128, 445-450 (2001).
41. Russell Kerr, Amber C. Kohl, Jennifer M. Boehnlein, “Bioactive Compounds from Bryozoans” in “Marine Biotechnolgy” Volume 6, Elsevier, 2001.
40. Russell Kerr; Biosynthesis of Marine Natural Products, in “Studies in Natural Product Chemistry”, Elsevier Science Publishers, Amsterdam; Vol. 21 pp 293-328 (2000).
39. Amber Coleman and Russell Kerr. Radioactivity-guided Isolation and Characterization of the Bicyclic Pseudopterosin Diterpene Cyclase Product from Pseudopterogorgia elisabethae. Tetrahedron, 56, 9569-9574 (2000).
38. Shanti Jeedigunta, Joann Krenisky and Russell Kerr. Diketopiperazines as advanced intermediates in the biosynthesis of ecteinascidins. Tetrahedron 56, 3303-3307 (2000).
37. Athar Ata and Russell Kerr. Elisabethamine: a new diterpene alkaloid from Pseudopterogorgia elisabethae. Tetrahedron Letters 41, 5821-5825 (2000).
36. Samina Naz, Russell Kerr and Ramaswamy Narayanan. New antiproliferative epoxysterols from Pseudopterogorgia americana. Tetrahedron Letters 41, 6035-6040 (2000).
35. Athar Ata and Russell Kerr. 12-Acetoxypseudopterolide: A New Diterpene from Pseudopterogorgia elisabethae. Heterocycles, 53, 717 (2000).
34. Amber C. Coleman, Laura Mydlarz and Russell Kerr. In Vivo and In Vitro Investigations into the Biosynthetic Relatedness of the Pseudopterosins. Organic Letters, 1, 2173-2175 (1999).
33. Paul Andrade, Robin Willoughby, Shirley Pomponi and Russell Kerr. Biosynthetic Studies of the Alkaloid, Stevensine, in a Cell Culture of the Marine Sponge Teichaxinella morchella. Tetrahedron Letters, 40, 4775-4778 (1999).
32. R.G. Kerr and S.S. Kerr; Marine Natural Products as Therapeutic Agents. Expert Opinion on Therpeutic Patents; 9, 1207-1222 (1999).
31. Russell Kerr, Richard Vicchiarelli and Stacey Kerr. Identification and biosynthetic origins of sterols in the marine bryozoan Bugula neritina. J. Nat. Prod. 62, 468-470, (1999).
30. Russell Kerr and Kathleen Kelly. An enzyme-based formaldehyde assay and its utility in a sponge sterol biosynthetic pathway. J. Nat. Prod. 62, 201-202, (1999).
29. Jens Knauer, Russell Kerr, David Lindley and Paul C. Southgate. Sterol Metabolism of Pacific Oyster (Crassostrea gigas) Spat. Comp. Biochem. and Physiol.,119B, 81 - 84 (1998).
28. Russell Kerr, Cathy Foss, Shigeki Matsunaga and Nobohiro Fusetani. Isolation and structure elucidation of epipolasterol and 22,23 dihydroepipolasterol from the marine sponge Epipolasis sp. Comp. Biochem. and Physiol.,117B, 5651-563 (1997).
27. Russell Kerr and Lesbeth Rodriguez. A chemoenzymatic production of 9(11)-secosteroids using and enzyme preparation of the gorgonian Pseudopterogorgia americana. In Vitro Cellular and Developmental Biology 33, 20A (1997).
26. Russell Kerr, Lesbeth Rodriguez and Jaelle Kellman. A chemoenzymatic synthesis of 9(11)-secosteroids using an enzyme extract of the marine gorgonian Pseudopterogorgia americana., Tetrahedron Letters. 37, 8301 (1996).
25. Russell Kerr, Joseph Lawry and Kim Gush. In vitro biosynthetic studies of the bryostatins, anti-cancer agents from the marine bryozoan Bugula neritina., Tetrahedron Letters. 37, 8305 (1996).
24. Xueshu Zhang, Ruiwen Zhang, Hui Zhao, Hongying Cai, Kimberly Gush, Russell Kerr, George Pettit and Andrew Kraft. Preclinical Pharmacology of the Natural Product Anticancer Agent Bryostatin 1, an Activator of Protein Kinase C. Cancer Research 56, 802 (1996).
23. Russell Kerr and Neil Miranda. Biosynthetic studies of ecteinascidins in the marine tunicate Ecteinascidia turbinata. J. Nat. Prod. 58, 1618 (1995).
22. Russell Kerr, Kathleen Kelly and Aaron Schulman. A novel biosynthetic route to pregnanes in the marine sponge Amphimedon compressa. J. Nat. Prod. 58, 1077 (1995).
21. Russell Kerr and Zhengjian Chen. In vivo and in vitro biosynthesis of saponins in Sea Cucumbers (Holothuroidea). J. Nat. Prod. 58, 172 (1995).
20. Russell Kerr and Michelle Kelly-Borges. Biochemical and morphological heterogeneity in the Caribbean sponge Xestospongia muta. Sponges in Time and Space, Proc. Int. Porifera Congress, 4th 65-73 (1994).
19. Russell Kerr, S.L. Kerr, Jane Formont, Martin Riddle and Peter Murphy. Chemotaxonomic relationships within, and comparisons between, the orders Haplosclerida and Petrosida (Porifera: Demospongiae) using sterol complements. Biochem. Syst. Ecol. 22, 735 - 52 (1994).
18. R.G. Kerr and P. Southgate; Isolation of gorgosterol from symbiont-free giant clam eggs and larvae. Comp. Biochem. and Physiol 104B, 707 (1993).
17. B.J. Baker and R.G. Kerr; Biosynthesis of Marine Sterols. in "Topics in Current Chemistry" 167, 1-31 (1993).
16. R.G. Kerr and B.J. Baker; Marine Sterols - A Review. Nat. Prod. Rep. 8, 465 (1992).
15. R.G. Kerr, S.L. Kerr, S. Malik and C. Djerassi; Biosynthetic Studies of Marine Lipids 38. Mechanism and Scope of Sterol Side Chain Dealkylation in Sponges; Evidence for Concurrent Alkylation and Dealkylation. J. Am. Chem. Soc. 114, 299 (1992).
14. R.G. Kerr, S.L. Kerr and C. Djerassi; Biosynthetic Studies of Marine Lipids 26. Elucidation of the Biosynthesis of Mutasterol, a Sponge Sterol with a Quaternary Carbon in its Side Chain. J. Org. Chem. 56, 63 (1991).
13. R.G. Kerr, S.L. Kerr, G.R. Pettit, D.L. Herald, T.L. Groy and C. Djerassi; Sterols of Marine Invertebrates 63. Isolation and Structure Elucidation of Sutinasterol, the Major Sterol of the Marine Sponge Xestospongia sp. J. Org. Chem. 56, 58 (1991).
12. R.G. Kerr, B.J. Baker, S.L. Kerr and C. Djerassi; Biosynthetic Studies of Marine Lipids 29. Demonstration of Sterol Side Chain Dealkylation Using Cell-Free Extracts of Marine Sponges. Tetrahedron Lett. 31, 5425 (1990).
11. R.G. Kerr, I.L. Stoilov, J.E. Thompson and C. Djerassi; Biosynthetic Studies of Marine Lipids 16. De Novo Sterol Biosynthesis in Sponges. Incorporation and Transformation of Cycloartenol and Lanosterol into Unconventional Sterols of Marine and Freshwater Sponges. Tetrahedron 45, 1893 (1989).
10. S. Malik, R.G. Kerr and C. Djerassi; Biosynthetic Studies of Marine Lipids 19. Dealkylation of the Sterol Side Chain in Sponges. J. Am. Chem. Soc. 110, 6895 (1988).
9. M.L. Cordeiro, R.G. Kerr and C. Djerassi; Biosynthetic Studies of Marine Lipids 15. Conversion of Parkeol (Lanost-9(11),24-dien-3b-ol) to 14a-methylcholest-9(11)-en-3b-ol in the Sea Cucumber Holothuria arenicola. Tetrahedron Lett. 2159 (1988).
Collaboration and Sponsorship
Research is currently sponsored by the following agencies.
 |
 The Jeanne and J.-Louis Lévesque Foundation
|
 |
Innovation PEI PEI
 |
|
The Atlantic Innovation Fund
 
|
|






















.jpg)

















